Product Description
Looking for 2 X the power of L-leucine? Then you need to try leucine peptides
Protein Maximizer: Leucine Peptides, 2 X more powder than L-leucine for greater muscle gains.
Turn your protein into a super muscle builder!
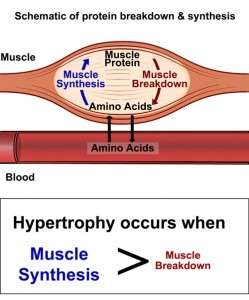
Protein Maximizer: Leucine Peptides delivers an influx of the power di and tri peptide leucine. Leucine, when supplemented, helps supports an increase in protein synthesis to build muscle.
For the ultimate in purity, Protein Maximizer: Leucine Peptides is unflavored.
- NO Salt
- NO Sugar
- NO Fillers
- NO Sucralose
- NO Aspartame
- NO Preservatives
- NO Acesulfame-K
- NO Artificial Colors
- NO Artificial Flavors
- NO Artificial Sweeteners
- NO Banned Substances
- NO caffeine or other stimulants
Proteinfactory.com® research studies on Leucine to build muscle.
Leucine Regulates Translation Initiation of Protein Synthesis in Skeletal Muscle after Exercise
Layne E. Norton and Donald K. Layman
Division of Nutritional Sciences, Department of Food Science and Human Nutrition, University of Illinois at Urbana-Champaign, Urbana, IL 61801
Koopman R, Wagenmakers AJ, Manders RJ, Zorenc AH, Senden JM, Gorselink M, Keizer HA, van Loon LJ. (2005) Combined ingestion of protein and free leucine with carbohydrate increases postexercise muscle protein synthesis in vivo in male subjects. Am. J. Physiol. Endocrinol. Metab. 288(4): E645-653
Merrick, W. C., & Hershey, J. W. B. (2000) The pathway and mechanism of initiation of protein synthesis. In: Sonnenberg N, Hershey JWB, Mathews MB, editors. Translational control of gene expression. Cold Spring Harbor Laboratory Press.*
Bolster, D. R., Crozier, S. J., Kimball, S. R., & Jefferson, L. S. (2002) AMP-activated protein kinase suppresses protein synthesis in rat skeletal muscle through down-regulated mammalian target of rapamycin (mTOR) signaling. J. Biol. Chem. 277: 23977-23980.
Take 1-2 teaspoons 15 to 30 minutes before training. And same thing post workout. Flavor as desired; mix with your favorite juice, or sports drink.
Benefits
Leucine and Whey may help increase insulin
Insulin is a powerful anabolic hormone. Equals huge muscle gains!!*
From what we have seen in research studies, drinking whey and leucine pre workout increases protein synthesis.
Leucine helps activates mTOR – to switch on muscle building
mToR plays an important part in building muscle.*
Leucine helps you lose weight
Some studies show that leucine helps reduce fat. *
These are based on L-leuince. Greater benefits with leucine peptides
More Details
Specifically using high quality protein such as whey protein contains an extraordinarily high amount of BCAA’s (over 30%). BCAA’s are known as the building blocks of muscle. BCAA’are made up of Leucine, Valine, and Isoleucine. Recently it has become known that taking 5 grams of Leucine with a protein shake or protein meal will increase protein synthesis by up to 70%. But the problem is that one can only buy L-Leucine, which is a single amino acid. This creates problems in the digestive tract as the body has trouble digesting single free form amino acids. Therefore, nutrition company’s and research groups that recommend using L-Leucine in hopes to increasing protein synthesis, instruct to take L-Leucine with food (other amino acids). But still this may not be enough for the body to digest and absorb the L-Leucine powder. To solve this problem, ProteinFactory.com is now offering Leucine Peptides. Leucine Peptides are in the form of di and tri peptides, or two or three amino acids linked together. The body absorbs amino acids through the intestinal tract and into the blood stream in the form of di and tri peptides. Our Leucine Peptides are made in the USA from the finest quality whey protein isolate and contains a complete amino acid profile including BCAA’s.
If taken 3 times per day 1 pound will last 30 days.
More Detailed Instructions: Mix 1 tsp of Leucine Peptides with your protein shake or sprinkle on any protein meal. Do not exceed 4 times per day. Best time to use Leucine Peptides is post workout with a hydrolyzed protein like (PeptoPro, or Hydrolyzed 520) and a Carbohydrate. For beginners or those concerned with taste, use Leucine Peptides with one of our Whey Protein Isolates, like Native Whey Isolate or Ultra Amino 97 Whey Isolate